This page looks at the role of the amygdala in emotional behaviour.
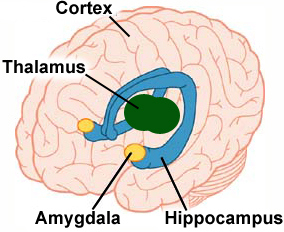
The amygdaloid complex comprises a structure of nuclei, deep within the temporal lobe of the brain (Sah, Faber, De Armentia, & Power, 2003). It plays an essential role in humans’ emotional perception of stimuli—particularly sensory information associated with fear or potential threat (Öhman, 2005). In short, this complex structure is the control centre of fear. There is also converging evidence to support the view that the amygdala plays a vital role in the development, and outward expression of, conditioned fear (Davis, 1992; Maren & Fanselow, 1996). In addition, it is also involved in survival—particularly in locating food and water (Carlson, 2013)—nurturing and protecting offspring (Fishbane, 2007; Zebrowitz, Luevano, Bronstad, & Aharon, 2009), emotional learning (Davis, 1994), the retrieval of emotional memories (Buchanan, 2007), visual awareness (Duncan & Barrett, 2007), decision-making (Bechara, Damasio, & Damasio, 2003), the processing of visual social stimuli (Adolphs & Spezio, 2006) and emotional behaviour generally.
Emotion response, in both humans and other complex vertebrates, involves a dynamic interplay between behavioural, autonomic and hormonal components (Carlson, 2013). These are all controlled by different neural systems and subsystems; however, fear, threat, survival instincts and our ‘emotional brain’ generally seem to be controlled by the amygdala. Described as the ‘quick and dirty route’ (LeDoux, 2000), evolutionary psychologists have stressed the importance of the amygdala in complex vertebrates, helping species to identify, and subsequently re-evaluate, potential threat situations. These events are received along the cortical pathway (LeDoux & Phelps, 1993). As the amygdala is essential to many animals for survival, it is not surprising, therefore, that many, if not all, the sensory modalities are utilized in the detection of potential emotional involvement—that is to say, neurons within the structure of the amygdala become active when emotionally-relevant stimuli are presented. For example, many researchers (LeDoux, Sakaguchi & Reis, 1984; LeDoux, Ruggiero & Reis,1985) have established that a single-tone conditioned stimulus (CS) when paired with an unconditioned stimulus (UCS)—specifically, a footshock—produces a fear reaction in rats. In line with computational modelling studies (Den Dulk, Heerebout & Phaf, 2003), this study indicates that animals can divert attention to potentially threatening situations in the wild, outside of their visual attention; thus, alerted, and in a state of readiness, the auditory cortex can assess the location of the noise to evaluate its potential threat. Indeed, in the clinical domain, researchers have pointed out that it is unexpected noise that often causes an initial fear response—particularly in PTSD sufferers (Walters & Hening, 1992). Adolphs et al (1999), in a study which looked at recognition of affect in human subjects with bilateral amygdala damage, concluded that the amygdala is extremely important in triggering information, particularly associated with threat and potential danger. Studies have also looked at how smell can influence emotional processing. Zald and Pardo (1997) measured regional cerebral blood flow (rCBF) in twenty healthy women using positron emission tomography; results from the study showed that aversive olfactory stimuli—specifically, a ‘sulfide cocktail’ containing dimethyl sulfide, ethanol, and methane—increased cerebral blood flow in the amygdala and left orbitofrontal cortex. And, Fontanini, Grossman, Figueroa and Katz (2009) provided strong evidence to support the view that, in rats, taste activates neurons in the basolateral amygdala.
Lesion studies in animals have been important in establishing the amygdala as an essential structure in the brain for registering and processing fear. Amaral (2003) showed that lesioned monkeys demonstrated a surprising lack of fear to snakes. More recently, Antoniadis, Winslow, David and Amaral (2007) found that selective ibotenic acid lesions in the amygdala of Rhesus monkeys—but, interestingly, not in the hippocampus—blocked ‘fear-potentiated startle’; however, in a second experiment, they found that animals which had learnt a fear-potentiated startle continued to show fear responses in spite of a complete lesion in the amygdala. This suggests that, although the amygdala is responsible for preliminary fear activation, it is not required for the overall expression, and preservation of, a fear startle. This supports the view of a number of researchers who have concluded that fear tends to be persistent. This has obvious evolutionary advantages in that animals can be alerted to potential threat on each occurrence; however, it can also cause unnecessary stress if the stimulus is no longer dangerous. In the laboratory, these fear reactions can be reduced by repeatedly pairing the CS without the negative UCS. In humans, the inability to cope with potential threats can lead to psychological disturbance including phobic anxiety, panic attacks and post traumatic stress disorder: treatments in clinical practice have tended to focus on behavioural or cognitive-behavioural modalities—for example, systematic desensitization and cognitive re-structuring (Chambless & Gillis, 1993; Deacon & Abramowitz, 2004).
It is also important to take into consideration the role of the social environment in fear. For example, in the earlier study (Amaral, 2003), a second experiment analysed fear responses in Rhesus monkeys who had received bilateral lesions of the amygdala at two weeks of age: consistently over a period of time, the monkeys showed no sign of distress when presented with snakes, and yet were fearful in new social situations. The author concluded that the amygdala was not important in the development of socially-specific knowledge and behaviour.
The order to which these emotional responses occur has been debated for many years. The James-Lange Peripheral Feedback Theory (Cannon, 1927) suggests that one first perceives and then, secondly, interprets potential danger, and that this affects the body and causes negative emotion; however, the Cannon-Bard Theory (Dalgleish, 2004) proposed that these changes—both physiological and emotional—happen simultaneously. But the amygdala is a complex structure: it sends impulses to the hypothalamus in order to activate the sympathetic nervous system ready for ‘fight or flight’ (Kloet, 2004; Papadimitriou & Priftis, 2009); the lateral nucleus receives sensory information from the neocortex, thalamus and hippocampus, and projects to the basal and central nucleus of the amygdala, while the central nucleus receives information from the basal and lateral nuclei. So, it is very difficult to generalize about its function, particularly as experiments have largely focussed on animals, and studies on humans have been, for the greater part, case studies.
But, if a fear reaction depends on context, it is important to investigate the connections between the amygdala and parts of the brain associated with the environment. Needlesstosay, a snake on the wildlife programme or a tiger in a zoo poses little threat; however, in a real live situation, it might understandably cause a fear response. Thus, conscious evaluation is an important consideration in emotional behaviour. The ventromedial prefontal cortex (VmPFC), for example, which connects with the hypothalamus and the amygdala, receives information about the environment and is also involved in the inhibition of emotional responses in various situations (Bremner et al.,1999). This mechanism supports Schachter’s Cognition-Plus-Feedback Theory (Schachter & Singer, 1962; Reisenzein, 1983). Further, researchers who have made experimental observations with regard to fear conditioning have suggested that malfunction in the medial prefrontal cortex make it extremely difficult for some patients to overcome their fears and anxieties (LeDoux, 1996). In addition, anxiety responses such as muscle tension, hyperhidrosis, accelerated heart rate beat or increased blood pressure may occur as a result of a mere association rather than potential or actual danger—for example, a particular smell or visual memory (Foa & Kozak, 1991). And yet, one of the most powerful techniques used in cognitive behavioural therapy (CBT) is one of re-framing in which the patient re-interprets the emotional content of a particular stimuli—for example, a driving scenario—in order to reduce and, hopefully, eliminate the fear response, as well as its concomitant psychosomatic features (Perczel-Forintos & Hackmann, 1999). Indeed, endocrine and autonomic responses are controlled by the central nucleus of the amygdala (Carlson, 2013), and dysregulation may give rise to prolonged periods of stress which, in turn, can have serious effects on health (Cohen & Herbert, 1996).
Unlike other animals, humans are able to think, make decisions, react and take the appropriate action: this is perhaps why both solution-focussed brief therapy and CBT, which focus on the patient’s control to make decisions, are so helpful in clinical practice. However, this action, certainly before therapy, might consist entirely of avoidance behaviour which may have a deleterious effect on well-being; indeed, the amygdala is utilized in avoidance learning, although is not required for long-term storage (McGaugh, Cahill & Roozendaal,1996).
It is hoped that a greater understanding of the role of the amygdala will help clinicians to gain a better understanding of psychological disorders. For instance, the amygdala also plays an important role in aggressive behaviour. For example, Trimble & Van Elst (1999) compared aggressive patients with a control group and found that a sub-group with aggressive tendencies had left-sided amygdala atrophy and a history of encephalitis. Stephens and Duka (2008), in a parallel design, found that human binge drinkers, alcoholics (who had undergone regular detoxication) and rodents, all of whom had repeatedly been denied access to alcohol, suffered reduced plasticity in the amygdala and hippocampus regions. Interestingly, converging evidence has pointed to the fact that the amygdala is important in the processing of rewards, as well as using rewards to motivate and reinforce behaviour (Cardinal, Parkinson, Hall & Everitt, 2002)—mechanisms which are no doubt inextricably interconnected with some impulsive behaviours. Further, it has been shown that individuals suffering from social anxiety, when presented with angry, harsh and fearful faces, displayed greater amygdala activation compared with controls (Phan, Fitzgerald, Nathan, & Tancer, 2006).
This report has analyzed the role of the amygdala in emotional behaviour. There has been a great deal of research in this area, but quantitative evidence is needed to sustain an understanding of the link between amygdala activation and clinical symptomatology. Further, although suitable subjects may be scarce, there needs to be further evidence that will investigate emotional behaviour in humans with amygdala damage.
References
Adolphs, R., & Spezio, M. (2006). Role of the amygdala in processing visual social stimuli. Progress in Brain Research, 156, 363-378.
Adolphs, R., Tranel, D., Hamann, S., Young, A. W., Calder, A. J., Phelps, E. A., & Damasio, A. R. (1999). Recognition of facial emotion in nine individuals with bilateral amygdala damage. Neuropsychologia, 37, 1111-1117.
Amaral, D. G. (2003). The amygdala, social behavior, and danger detection. Annals of the New York Academy of Sciences, 1000, 337-347.
Antoniadis, E. A., Winslow, J. T., Davis, M., & Amaral, D. G. (2007). Role of the primate amygdala in fear-potentiated startle: effects of chronic lesions in the rhesus monkey. The Journal of Neuroscience, 27, 7386-7396.
Bechara, A., Damasio, H., & Damasio, A. R. (2003). Role of the Amygdala in Decision‐Making. Annals of the New York Academy of Sciences, 985, 356-369.
Bremner, J.D., Staib, L.H., Kaloupek, D., Southwick, S.M., Soufer, R. & Charney, D.S. (1999). Neural correlates of exposure to traumatic pictures and sound in Vietnam combat veterans with and without post-traumatic stress disorder: a positron emission tomography study. Biological Psychiatry, 45,806–816.
Buchanan, T. W. (2007). Retrieval of emotional memories. Psychological Bulletin, 133, 761-779.
Cannon, W.B. (1927). The James-Lange theory of emotions: A critical examination and an alternative theory. American Journal of Psychology, 39, 106-124.
Cardinal, R. N., Parkinson, J. A., Hall, J., & Everitt, B. J. (2002). Emotion and motivation: the role of the amygdala, ventral striatum, and prefrontal cortex. Neuroscience and Biobehavioral Reviews, 26, 321-352.
Carlson, N.R. (2013). Physiology of Behavior (11th edition). Boston: Pearson.
Chambless, D. L., & Gillis, M. M. (1993). Cognitive therapy of anxiety disorders. Journal of Consulting and Clinical Psychology, 61, 248-248.
Cohen, S., & Herbert, T. B. (1996). Health psychology: Psychological factors and physical disease from the perspective of human psychoneuroimmunology. Annual Review of Psychology, 47, 113-142.
Dalgleish, T. (2004). The emotional brain. Nature Reviews Neuroscience, 5, 583-589.
Davis, M. (1992). The role of the amygdala in fear and anxiety. Annual Review of Neuroscience, 15, 353-375.
Davis, M. (1994). The role of the amygdala in emotional learning. International Review of Neurobiology, 225-225.
Deacon, B. J., & Abramowitz, J. S. (2004). Cognitive and behavioral treatments for anxiety disorders: a review of meta‐analytic findings. Journal of Clinical Psychology, 60, 429-441.
Dulk, P. D., Heerebout, B. T., & Phaf, R. H. (2003). A computational study into the evolution of dual-route dynamics for affective processing. Journal of Cognitive Neuroscience, 15, 194-208.
Duncan, S., & Barrett, L. F. (2007). The role of the amygdala in visual awareness. Trends in Cognitive Sciences, 11, 190-192.
Foa, E. B., & Kozak, M. J. (1991). Emotional processing: theory, research, and clinical implications for anxiety disorders. In J. D. Safran & L. S. Greenberg (Eds.), Emotion, psychotherapy, and change (pp. 21–49). New York: Guilford Press.
Fontanini, A., Grossman, S. E., Figueroa, J. A., & Katz, D. B. (2009). Distinct subtypes of basolateral amygdala taste neurons reflect palatability and reward. The Journal of Neuroscience, 29, 2486-2495.
Kloet, E. R. (2004). Hormones and the stressed brain. Annals of the New York Academy of Sciences, 1018, 1-15.
LeDoux, J. (1998). The emotional brain: The mysterious underpinnings of emotional life. New York: Simon and Schuster.
Ledoux, J. (2000). Cognitive-emotional interactions: Listen to the brain. Cognitive neuroscience of emotion, 129-155.
LeDoux, J. E., & Phelps, E. A. (1993). Emotional networks in the brain. In M Lewis and JM Haviland-Jones (Eds) Handbook of Emotions (pp109-118). New York: Guilford.
LeDoux, J. E., Ruggiero, D. A., & Reis, D. (1985). Projections to the subcortical forebrain from anatomically defined regions of the medial geniculate body in the rat. Journal of Comparative Neurology, 242, 182-213.
LeDoux, J. E., Sakaguchi, A. K. I. R. A., & Reis, D. J. (1984). Subcortical efferent projections of the medial geniculate nucleus mediate emotional responses conditioned to acoustic stimuli. The Journal of Neuroscience, 4, 683-698.
Maren, S., & Fanselow, M. S. (1996). The amygdala and fear conditioning: Has the nut been cracked? Neuron, 16, 237–240.
McGaugh, J. L., Cahill, L., & Roozendaal, B. (1996). Involvement of the amygdala in memory storage: interaction with other brain systems. Proceedings of the National Academy of Sciences, 93, 13508-13514.
Öhman, A. (2005). The role of the amygdala in human fear: Automatic detection of threat. Psychoneuroendocrinology, 30, 953-958.
Papadimitriou, A., & Priftis, K. N. (2009). Regulation of the hypothalamic-pituitary-adrenal axis. Neuroimmunomodulation, 16, 265-271.
Perczel-Forintos, D., & Hackmann, A. (1999). Transformation of Meaning & its Effects on Cognitive Behavioural Treatment of an Injection Phobia. Behavioural and Cognitive Psychotherapy, 27, 369-376.
Phan, K. L., Fitzgerald, D. A., Nathan, P. J., & Tancer, M. E. (2006). Association between amygdala hyperactivity to harsh faces and severity of social anxiety in generalized social phobia. Biological Psychiatry, 59, 424-429.
Reisenzein, R. (1983). The Schachter theory of emotion: Two decades later. Psychological Bulletin, 94,239-264.
Sah, P., Faber, E. S. L., De Armentia, M. L., & Power, J. (2003). The amygdaloid complex: anatomy and physiology. Physiological Reviews, 83, 803-834.
Schachter, S & Singer, J.E. (1962). Cognitive, social, and physiological determinants of emotional state. Psychological Review, 69,379-399.
Stephens, D. N., & Duka, T. (2008). Cognitive and emotional consequences of binge drinking: role of amygdala and prefrontal cortex. Philosophical Transactions of the Royal Society B: Biological Sciences, 363, 3169-3179.
Trimble, M. R., & Elst, L. T. (1999). On some clinical implications of the ventral striatum and the extended amygdala: Investigations of aggression. Annals of the New York Academy of Sciences, 877, 638-644.
Walters, A. S., & Hening, W. A. (1992). Noise‐induced psychogenic tremor associated with post‐traumatic stress disorder. Movement Disorders, 7, 333-338.
Zald, D. H., & Pardo, J. V. (1997). Emotion, olfaction, and the human amygdala: amygdala activation during aversive olfactory stimulation. Proceedings of the National Academy of Sciences, 94, 4119-4124.
David Kraft PhD


